 While some companies have failed in their endeavours, there is still a strong and thriving pipeline of innovation for adapted and new vaccines. This past year has demonstrated industry’s ability to be agile and respond to new challenges but has also underlined the importance of pursuing innovation. Addressing vaccine hesitancy requires collective focus as there continue to be pockets of people that, after 18 months of safety data and over 8 billion doses administered around the planet, remain hesitant towards COVID-19 vaccines.Regulatory approval even before doses reach countries, requires the World Health Organization (WHO) and national regulatory authorities to work together to ensure that the vaccines are approved at national level, so that doses can be deployed immediately.Extension of vaccine shelf life, with the increase in dose-sharing and distribution of vaccines, coupled with the challenges related to country absorption, innovation and regulatory oversight is needed so that shelf life is extended, in line with ongoing studies looking at the stability of vaccines, to ensure vaccines are not wasted.Political prioritization, funding, infrastructure and human resources must be sufficient to deliver vaccines safely, including upscaling cold chain capabilities from airfields to the last mile and increasing health workforce numbers to deliver the vaccines even in areas that are hard to reach, and without compromising routine immunization.Attention is urgently needed to focus on concrete measures in recipient countries to support COVID-19 vaccine deployment and uptake: 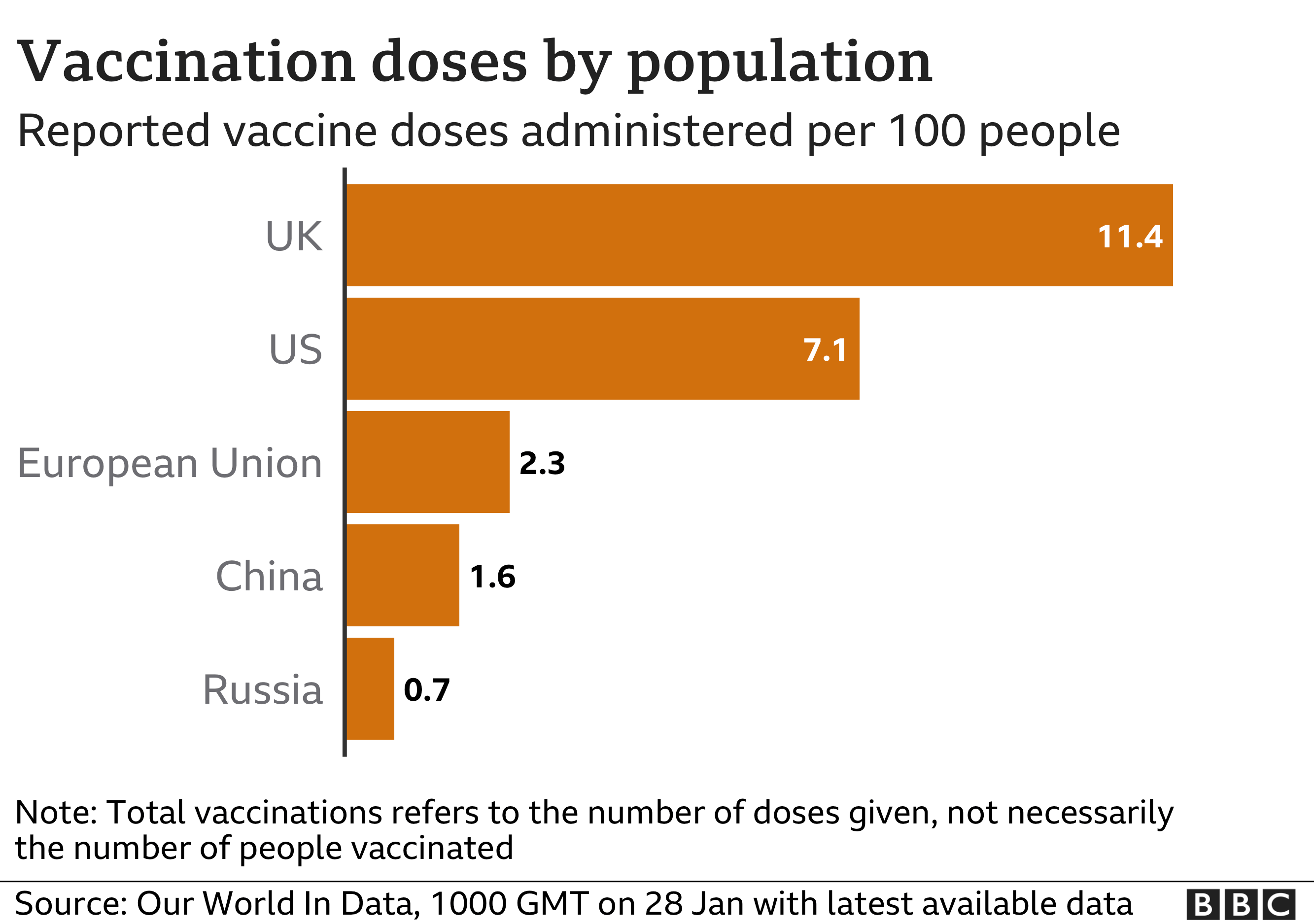
Rapid and efficient delivery of COVID-19 vaccines, inclusive of dose-sharing, requires effective and flexible coordination and planning. Today, more than 700 million doses have been shipped by COVAX to 144 countries and nearly 1 billion doses ordered. 
Independent analysis by Airfinity has shown that, by the end of March 2022, G7 and EU countries are projected to have 1,4 billion surplus vaccines, even when administering boosters.ĭose-sharing is gaining momentum to reach those who have not yet been vaccinated, and COVAX is ramping up fast to ensure doses are distributed equitably around the world. The 2021 supply of COVID-19 vaccines resulted in half of the world’s population being vaccinated within a year. Of these deals, 229 include various forms of voluntary collaboration that rely on technology transfer, sharing of know-how about the processes and the technologies used to make the vaccines, as well as training specialist personnel to ensure quality standards. The manufacturing scale up of COVID-19 vaccines that had been developed in record time required building new production lines able to consistently produce millions of doses to the highest quality standards, managing global supply chains for hundreds of components and ingredients, as well as forging over 300 partnerships around the world to increase manufacturing output. Today, with a surge in doses from the international partnership set up to distribute vaccines to lower-income countries, called COVAX, as well as bilateral arrangements, the focus must now be on reducing the time between arrival of vaccines to vaccinations. Supplies have been distributed unevenly, but countries have started to release surplus doses so that they can be shared. The Biotechnology Innovation Organization (BIO), Developing Countries Vaccine Manufacturers’ Network (DCVMN), and the International Federation of Pharmaceutical Manufacturers and Associations (IFPMA), representing many of the companies behind the historic scale up of manufacturing of vaccines, released independent data that confirms production of COVID-19 vaccines scaled up from zero to 11,2 billion doses in just one year. #AlwaysInnovating: The pharmaceutical innovation journey.Upholding ethics and business integrity.Quality, safety, and efficacy of therapeutical products. 
Collaboration, convergence, and regulatory reliance.Improving health security and pandemic preparedness.Fostering a thriving innovation ecosystem. 
0 Comments
Leave a Reply. |

 RSS Feed
RSS Feed
